Clinical Transfer Imaging - Activities
The clinical transfer team of the Imaging and Cytometry Platform is part of the Surgery and Pathology Photonic Imaging Group-SP2I
Gustave Roussy's Surgery and Pathology Photonic Imaging Group-SP2I apply photonic imaging technologies to help clinicians solve problems in surgery and pathology for which other conventional imaging modalities currently offer no solution. Photonic imaging refers to all imaging modalities based on the interaction of light with tissue. Advances in optics, instrumental engineering (miniaturization, sensor sensitivity, etc.) and molecular chemistry have made it possible to implement photonic imaging in clinical practice as new exploratory protocols, ultimately improving patient care. Minimally invasive, photonic imaging provides real-time information in the operating room or in pathology.
SP2I's major asset is the close and well-established collaboration between photonic imaging specialists, pathologists and surgeons for:
- the development of innovative methods incorporating photonic imaging to set up new protocols, potential future standards at Gustave Roussy
- the development of new methodologies to improve key stages in patient management in surgery and pathology, such as tissue or organ identification (resection margin control, biopsy diagnosis, parathyroid gland preservation...) and functional information (flap viability, lymphatic drainage...).
- new approaches that can be applied to clinical routine in the short term, or on the clinical validation of prototypes supplied by academic laboratories or industry.
SP2I group is a member of the scientific consortium of INSERM's IMPULSION program "Surgical and interventional procedures of the future".
Since 2008, over 1,300 patients have benefited from innovative imaging technologies in various clinical studies:
- Preclinical study followed by a bicentric clinical study (PI: Ingrid Breuskin) on the use of confocal endomicroscopy in head and neck cancers - MEC-ORL project
Funding source: INCa, la Fondation des « Gueules Cassées », fondation Gustave Roussy, PHRC-K
Bicentric clinical study with Toulouse IUCT Oncopôle (Pr S. Vergez)
NCT02994225 - Preclinical study to develop endomicroscopy for the diagnosis of peritoneal carcinosis - PERSEE project.
Funding source: BPI France - European preclinical study to develop and transfer full-field optical coherence tomography for head and neck cancer - CAREIOCA project.
Funding source: financement européen
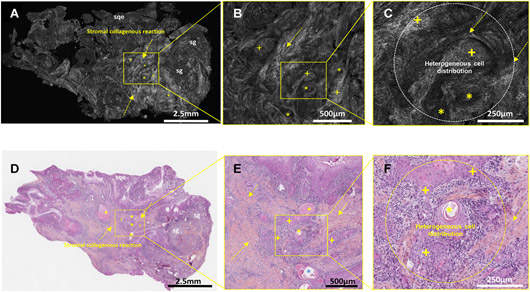
- Preclinical and clinical study of parathyroid gland preservation in thyroid cancer surgery using near-infrared autofluorescence macroscopy (PI Dana Hartl)
Funding source: Ligue contre le cancer - Clinical study on the identification of sentinel lymph nodes by near-infrared fluorescence macroscopy in breast cancer surgery (PI Chafika Mazouni)
- Clinical study of flap perfusion assessment using near-infrared fluorescence imaging in head and neck reconstructive surgery (PI Quentin Qassemyar)
Funding source: Industriel - Clinical study on axillary reverse mapping in breast cancer surgery using near-infrared fluorescence (PI Chafika Mazouni)
Funding source: PHRC-K
NCT02994225 - Clinical study on the evaluation of tumor boundaries after indocyanine green administration in NIR 1 head and neck cancer (PI Philippe Gorphe) - MAGNOLIA project
Funding source: PHRC-K
Bicentric clinical study with the Jules Bordet Institute in Brussels (Dr A.Digonnet)
NCT04842162 - Ultra-fast, wide-field confocal microscopy for breast cancer lumpectomy imaging (PI Angelica Conversano & Marie-Christine Mathieu) - HIBISCUSS project
Funding source: Industriel
NCT04976556 - Ultra-fast, wide-field confocal microscopy for real-time diagnosis of sentinel lymph nodes ex vivo in head and neck cancer (PI Ingrid Breuskin) - HISTOLOG-ORL project
Funding source: Fondation de l'avenir et La Société Française d’Oto-Rhino-Laryngologie et de Chirurgie de la Face et du Cou.
- Breast carcinoma detection by ex vivo fluorescence confocal microscopy for one-stop breast clinic workflow ((PI Marie-Christine Mathieu) – LYRIC project
- Ex vivo confocal microscopy for imaging resection margins in ENT cancer (PI Odile Casiraghi & Muriel Abbaci) – CARINA project
- Diagnostic accuracy of ultra-fast confocal imaging for intraoperative control of the retroareolar border of mastectomies with preservation of the skin sheath (PI Angelica Conversano) – CAMELIA project
Funding source: Fondation de l'avenir - Fluorescence imaging coupled with the specific marker emi-137 for intraoperative control in cancer surgery (PI Muriel Abbaci, Ingrid Breuskin, Isabelle Sourrouille, Angelica Conversano) – IMPULSION project
- Ancillary study: Spidermass technology for the detection of head and neck cancers and peritoneal metastases – SPIDER-IMPULSION project in collaboration with Prof. Isabelle Fournier, PRISM laboratory - Inserm U1192
- Ancillary study: Fluorescence signal quantification in surgery: camera versus fiber spectroscopy – SPECTRA-IMPULSION project in collaboration with Prof. Bruno Montcel, CREATIS laboratory – Inserm U1294
Funding source: Booster program INSERM Procédures chirurgicales et interventionnelles du futur - Preoperative identification of perforators by photoacoustic tomography for reconstructive surgery of head and neck cancers- IMAGO project (Nadia Benmoussa-Rebibo)
Funding source: Ligue contre le cancer
Our past and present INDUSTRIAL COLLABORATIONS include Proimaging, Avelas Biosciences,Biospace, Clerad, Kaer Labs, LLtech, Mauna Kea Tech, Quest medical imaging, SamanTree medical, Vivascope GmbH, Edinburgh Molecular Imaging, Serb, Deep Color imaging.
PATENT : red and far-red fluorescent dyes for the characterization of biological tissues at a cellular level fr 27452/024WO1
Small animal imaging
From whole body…
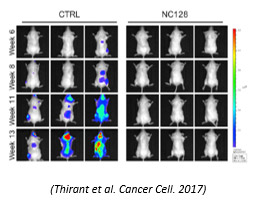
Noninvasive imaging is a major tool and became essential for evaluating tumor models. Current strategies based on deep orthotopic grafts require imaging tools for qualitative and quantitative evaluation in situ without invasive interventions affecting biology. Bioluminescence (BLI)/fluorescence (FLI) or X-ray microtomography imaging meets these requirements. These technologies, available to research teams, are located at the PFIC platform for preclinical research: IVIS Lumina III/IVIS Spectrum (BLI/FLI imaging), Quantum GX3 (µCT).
These instruments allow:
- focused or whole body imaging in real-time for a sensitive and early detection of primary tumors and metastases
- long-term, qualitative and quantitative studies on same individuals
- combination of biologic informations simultaneously collected from multiple bioluminescent/fluorescent probes (IVIS LuminaIII and SpectrumCT) (max 5 biomarkers simultaneously).
The IVIS Spectrum also allows combination of CT tomographic imaging with optical imaging, enabling the 3D localization of light sources detected by fluorescence or bioluminescence. For higher resolution X-ray tomographic imaging, the QuantumGX3 will be preferred. Fully configurable (X-ray source settings, FOV, scan speed and mode), it is adaptable for longitudinal in vivo whole-body imaging up to high-resolution ex vivo imaging. Its use is particularly indicated for imaging bone structures or soft tissues (lungs, liver, kidneys, etc.) that naturally have high X-ray contrast or through the administration of contrast agents.
Main applications of optical imaging conducted at Gustave Roussy are schematically summarized below:
- Dynamic characterization of tumor models and alternative preclinical models
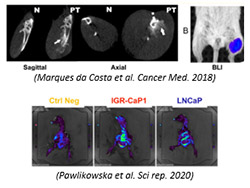
- Bioluminescence and fluorescence imaging
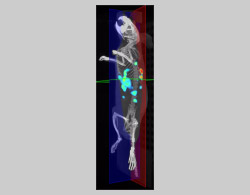
- Evaluation of therapeutic strategies (chemotherapy, vaccination, radiotherapy...) and new vectors of molecules
- Evaluation of new fluorescent contrast agents for early and specific detection of tumor lesions after systemic, subcutaneous or topic administration

In collaboration with academic research units or industrial groups, protocols have been developed in other clinical areas (examples: optimization of chemical composition for mucoadhesive gels, evaluation of a calcium channel promoter…)
To cell…
Whole body optical imaging is a powerful and highly sensitive tool adapted to multiple individuals screening. However, macroscopic evaluation needs to be completed/explained by the underlying cellular mechanisms. High resolution in vivo imaging at cellular level is achieved on isolated organs or through dorsal skinfold chamber both allowing visualization of cells/microenvironment interactions. These techniques associated with a macroscope (AZ100M, Nikon) or a confocal/multiphoton (SP8, Leica) allow to characterize in vivo :
- tissue and vessel architecture and extra/intra hemodynamics
- tumor dynamics and metastatic spreading
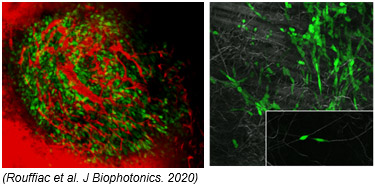
- cellular interactions
- evaluation of therapeutic efficacy
Our intravital imaging technologies are also available to the national scientific community thus leading to various protocols: 1- micromanipulation by thermal ablation and evaluation of tissue regeneration after injection of vascular progenitors (collab. U1197) ; 2- oxidative stress quantification during inflammation (collab. CNRS UMR 7645) ; 3- in vivo biodistribution of magnetoliposomes (collab. UMR8612) ; 4- dystrophin expression in macular degeneration (collab. END-ICAP UVSQ).
Closed to the preclinical experimentation platform, all in vivo procedures are conducted by a trained and authorized staff in compliance with European directives.
